NORD, the American National Organization for Rare Disorders, held its annual conference on 16 and 17 October 2017 in Washington DC.
Becky Gallis participated in this conference on behalf of CMTC-OVM US, and Lex van der Heijden did so on behalf of CMTC-OVM Nederland. We have been participating in this conference since 2004, and thanks to the treacle waffles and chocolate, our organisation is very well known! Here is a brief report of the 2017 edition.
The conference was well attended again this year, with 670 visitors. It offered us many networking opportunities; for example, we met Dr. Ramaiah Muthyala, chairman of the Indian Organization for Rare Diseases, who had our information leaflets translated into Hindi and Telugu. He is also going to promote our organization in India, with the aim of helping people who suffer from CMTC and other vascular malformations. About 100 million people in India suffer from a rare disease.
Complex situation in the USA due to regularly changing policy
Peter Saltonstall, CEO & President of NORD, opened the conference. One of the things he outlined, was the complex situation in the USA where the situation could change each day due to the current government’s policy. Peter indicated that 7,000 rare diseases are known at this moment and that 1 in 10 Americans suffers from any of those, i.e. about 32 million rare disease sufferers overall. In the Netherlands, 1 in 17 people suffer from a rare disease, which is about 1 million).
The Mighty
Mike Porath spoke about the whys and wherefores of the website ‘The Mighty.’ Mike, whose own child suffers from a rare disease, is the founder of The Mighty (www.themighty.com). He shared his personal story with us, including videos. The purpose of The Mighty is to share real stories of people who face major challenges because of – rare – diseases; issues that are not only of a medical but also of a psychological and psychosocial nature. By now, the Mighty has grown to an audience of 3 million people who read stories and watch video content. (We are part of this worldwide network as well and regularly publish articles from The Mighty on our Facebook page.) Through this network, we could also promote our organization worldwide and reach out to more patients and families, with the ultimate goal of helping patients and families.
FDA
Scott Gottlieb of the American Federal Drug Administration (FDA) discussed the authorization of (orphan) medicines. The FDA is an important organization in the USA, as they are responsible for the authorisation of medicines for the American market. Currently, the percentage of authorised orphan drugs (medicines for rare diseases) has increased to 70% of the applications. About 600 orphan drugs have been authorised at this moment, while about 7,000 rare diseases have been defined.
Tips for collaboration between patient organizations and industry
In the session ‘Ethical Guidelines for Patient Organizations & Industry to Collaborate’, Scott Gottlieb showed clearly and practically what patient organizations should focus on when collaborating with the industry. He gave the following tips:
- Beware of conflicting roles.
- Beware of conflicts of interests.
- Ensure that internal policies and procedures have been drawn up for this.
- Indicate your wishes as an organization at meetings with the industry; be transparent.
- Present what you do as an organization at meetings with the industry and ask your counterparts to do so as well.
- Communicate about such meetings in public (website, newsletters, etc.).
- Always keep in mind what is best for the patients – that is what it all comes down to in the end.
Overmedication
The session ‘Assuring Patient Access: Future Outlook for Patient Assistance Programs’ included parents’ deliberate medical abuse of their children by, for example, overmedication. In the USA, the child is placed into custody immediately in cases when this is observed. Unfortunately, errors of judgement sometimes occur, resulting in the unjust removal of children from their homes.
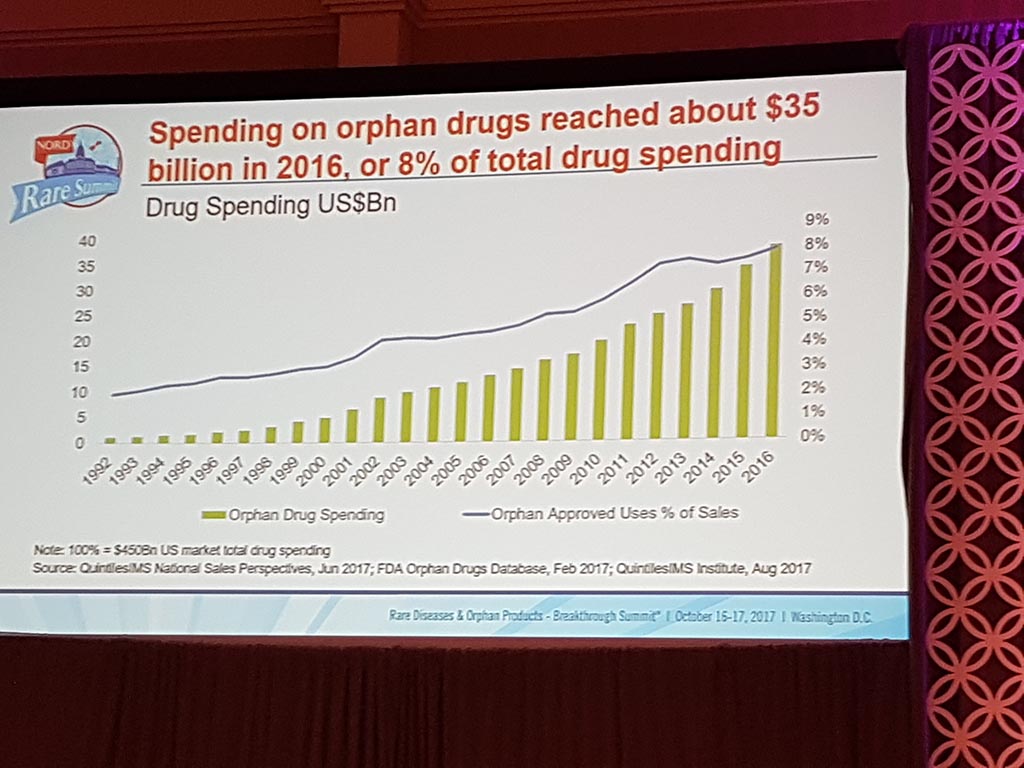
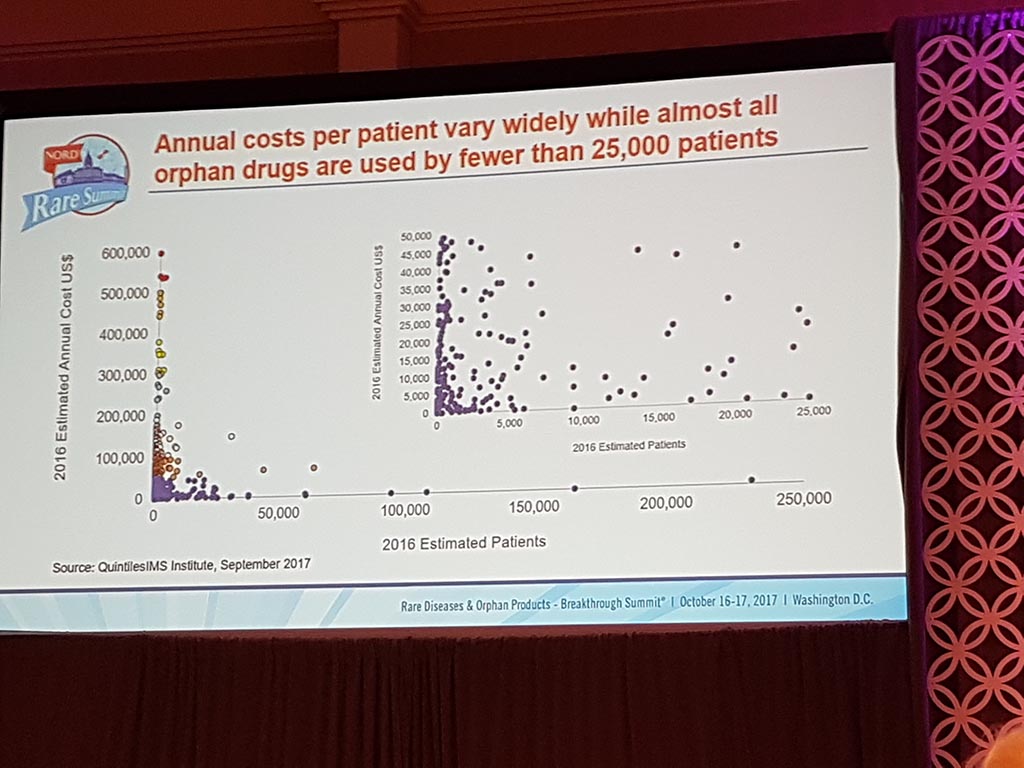
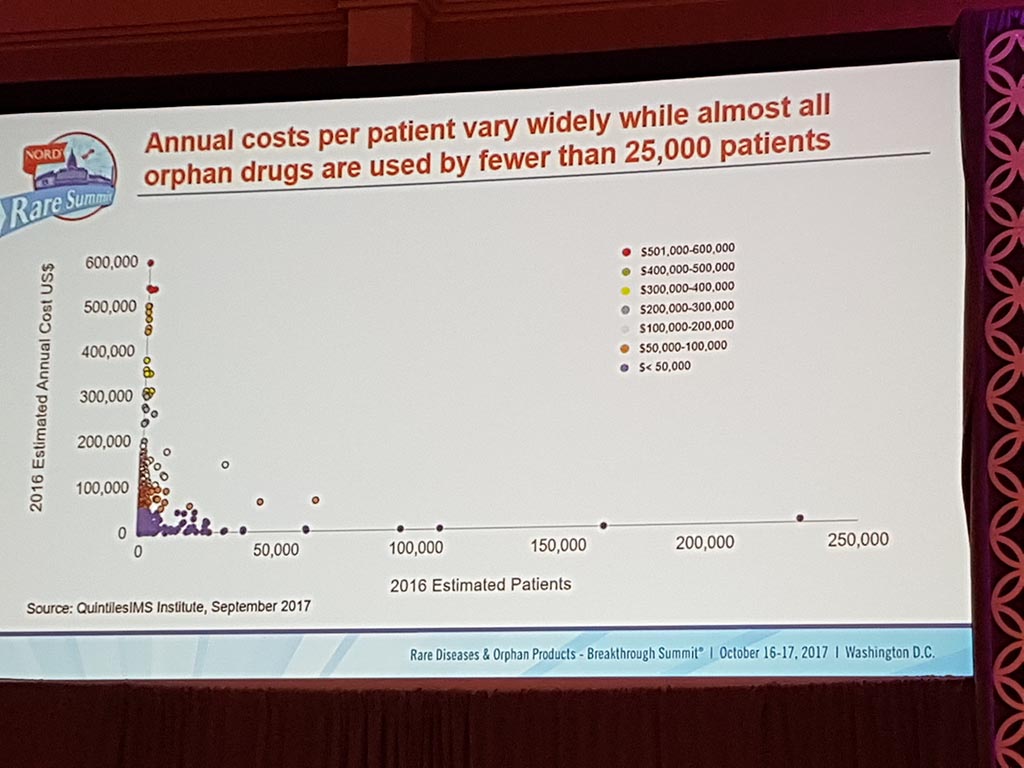
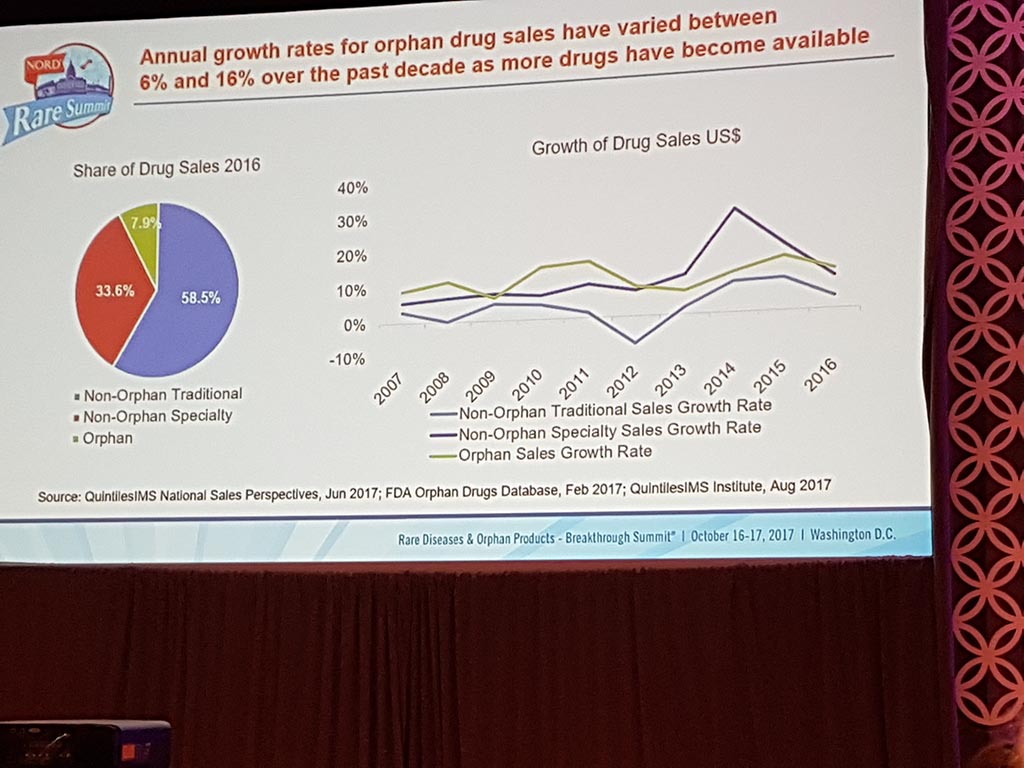
Extreme prices of medicines
The session ‘Challenges of Healthcare Costs & Treatment Prices’ showed that there are considerable issues with access to medication for patients in the USA as well, particularly in the case of life-saving medicines. Extreme prices are charged with here as well, especially if pharmaceutical companies have a monopoly position.
The importance of sharing medical data
The importance and effect of sharing medical data were emphasized in the session ‘The Power of Data Sharing’. Unfortunately, several hospitals and other healthcare providers in the USA seem not to have organized the storage and availability of medical data in such a way that these are available to patients. This is an issue in many countries.
Data in graphs showed that the life expectancy of patients suffering from many different types of diseases has increased enormously, thanks to the sharing of medical data. As a result, many examinations in patients can be prevented; after all, it makes no sense to repeat examinations when their outcome is already known. The prevention of double examinations also saves costs. One of the speakers also stated that all-important studies and results should be published via PubMed, so that these data will be made publicly available automatically after one year, and other researchers can reuse this data.
One of the topics discussed during a work lunch was ‘The Transition of Care from Child to Adult’. The major question was whether this has been arranged, and if so: how? Our organisation will focus on this subject and explore and elaborate on it in more detail.
Also Steve Groft participated again. He is the founder of the Office of Rare Diseases Research of the National Institute of Health (www.nih.gov), and was its director for over 30 years. Although officially, he has been retired for years, he ‘just’ doesn’t quit. We are proud of him being our ambassador!